Science Education Topics
Want to learn more about the latest advances in medical technology?
Featured Highlights
Explore new educational materials about biomedical engineering and imaging. Read inspiring profiles of researchers in biomedical engineering and imaging.
Biomedical engineering lesson plans that spark excitement and interest in middle schoolers and focus on how a biomedical engineering approach can be used to address health care problems. Download for free.
DEBUT challenges undergraduate student teams to develop technology solutions to unmet needs in any area of health care.
Test your knowledge on different types of medical imaging and learn how these technologies help clinicians see inside the human body.
Featured Videos
Researchers from chemical engineering, biomedical and bioengineering, biochemistry, and biophysics give their advice to science students and those who are thinking of going into science.
This short video briefly highlights 5 new cool technologies that your tax dollars have helped to fund.
NIBIB-funded researchers are working on a prosthetic ankle that uses the wearer's residual muscles - and the electrical signals that they generate - to help amputees with their postural stability.
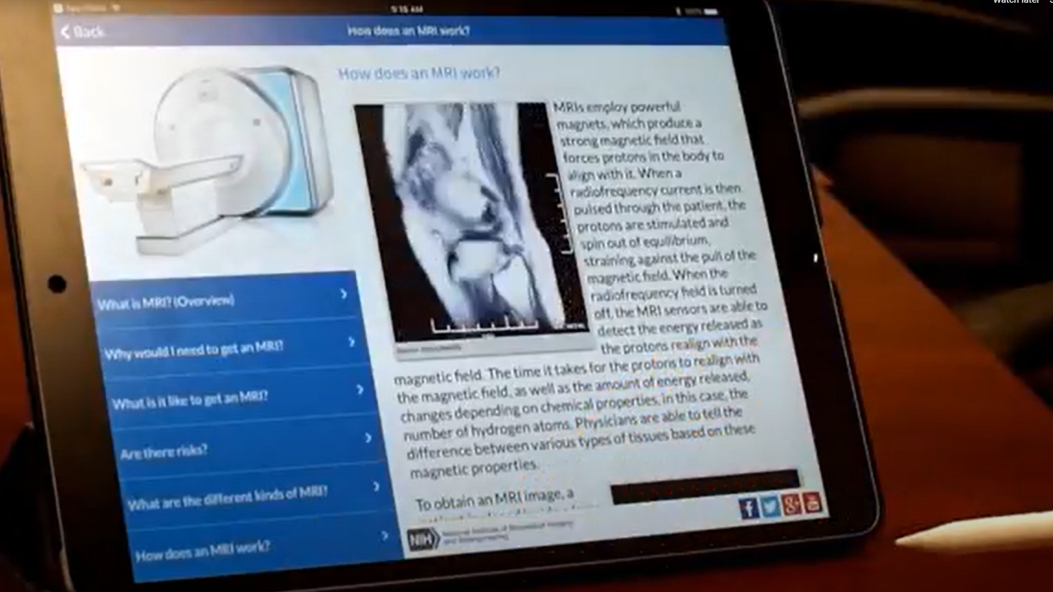
Medical Imaging App
Learn how medical scans work and what to expect during your next one. The app explains 5 different types of scans and describes the latest research in those modalities.



