- What is optical imaging?
- What types of optical imaging are there and what are they used for?
- What are the advantages of optical imaging?
- What are NIBIB-funded researchers developing in the area of optical imaging to improve biomedical research and medical care?
What is optical imaging?
Optical imaging uses the unique properties of light to obtain detailed medical images. When light interacts with objects, it can be absorbed, scattered, reflected, or even emitted. Scientist can leverage these interactions to visualize biological specimens, ranging from molecules and cells to entire tissues and living organisms.
The use of optical imaging in the health care space dates back hundreds of years, as physicians used microscopes as early as the 17th century. Microscopy is a foundational tool that still plays an important role in medicine today. This technique uses light and a system of lenses to magnify objects to hundreds if not thousands of times their original size. Pathologists rely on microscopy images to diagnose disease and guide treatment decisions, and researchers use the information generated from optical microscopy to learn about biological processes, which can lead to new discoveries.
Beyond microscopy, scientists can use a variety of different optical imaging techniques to visualize organs inside the body in a minimally or non-invasive way.
What types of optical imaging are there and what are they used for?
Optical imaging can help scientists to visualize a wide range of biological specimens, from something as small as DNA strands to something as large as an entire human organ.
Nanoscopic imaging allows researchers to see objects in the nanometer range, including DNA strands, proteins, and viral particles. Microscopic imaging enables scientists to visualize objects in the micrometer range, such as bacteria, red blood cells, and other cellular and subcellular structures. Macroscopic imaging allows clinicians to generate images of larger tissue samples, entire organs, entire animal models, or other specimens ranging from the millimeter to meter scale. Many of these techniques can be used to obtain non-invasive medical images of tissues and structures inside of the human body. Some examples of macroscopic imaging are detailed below.
Optical coherence tomography (OCT) is a technique for obtaining sub-surface images of structures, such as diseased tissue, just below the skin. OCT imaging is similar to ultrasound imaging, but instead of using sound waves and their resulting reflections to visualize tissues inside of the body, researchers use infrared light instead. Ophthalmologists use OCT to obtain detailed images from within the retina, and cardiologists also use this technique to help diagnose coronary artery disease.
Photoacoustic imaging is a hybrid imaging technique that combines optical techniques with ultrasound imaging to generate high-resolution images of tissues deep in the body. It works by delivering laser pulses to a patient’s tissues which absorb and generate ultrasound waves that can be detected with a specialized transducer. The technique can help monitor blood vessel growth in tumors, detect skin melanomas, and track blood oxygenation in tissues.
Diffuse optical tomography (DOT) and diffuse optical imaging (DOI) are non-invasive techniques that use light in the near-infrared region to measure tissue properties such as total hemoglobin concentration and blood oxygen saturation. Because DOT and DOI work well in soft tissue, the techniques are widely used for breast cancer imaging, brain functional imaging, stroke detection, photodynamic therapy, and radiation therapy monitoring.
Endoscopy methods can adapt similar optical imaging techniques above, such as microscopy-based techniques, utilizing fiber optics and miniature cameras to enable imaging of internal cavities and organs, such as the stomach. This approach allows the application of advanced optical imaging modalities within the body, making it possible to visualize and analyze tissues in their natural environment with high precision.
What are the advantages of optical imaging?
Optical imaging is particularly useful for measuring multiple properties of soft tissue. Because of the wide variety of ways different soft tissues absorb and scatter light, optical imaging can measure changes that are early markers of abnormal functioning of organs and tissues.
Importantly, optical imaging can be combined with other imaging techniques, such as MRI, to provide enhanced information for doctors monitoring complex diseases or researchers working on intricate experiments. Combinations of different optical imaging techniques also provides more detailed information about tissue at different scales.
Further, because visible, ultraviolet, and infrared light are less hazardous than other types of radiation, optical imaging techniques can be used for repeated procedures to monitor the progression of disease or the results of treatment.
What are NIBIB-funded researchers developing in the area of optical imaging to improve biomedical research and medical care?
Photoacoustic imaging to analyze blood flow: The qualities of flowing blood, or hemodynamics, hold important insights into vascular diseases, but technological limitations have largely kept measurements of these properties out of reach in the clinic. NIBIB-funded researchers have developed a low-cost, non-invasive 3D imaging method, called Photoacoustic Computed Tomography through an Ergodic Relay (PACTER), which is capable of imaging flowing blood in real time. Using their technology, the researchers could visualize blood vessels and calculate blood flow speeds in human hands and feet, which are commonly assessed in patients with vascular disease and diabetes. Their system could also non-invasively generate 3D maps of abdominal vasculature in mice. The researchers hope that that their method could someday be applied to measure blood oxygenation levels in the arteries of the neck and to track metabolism in the brain.
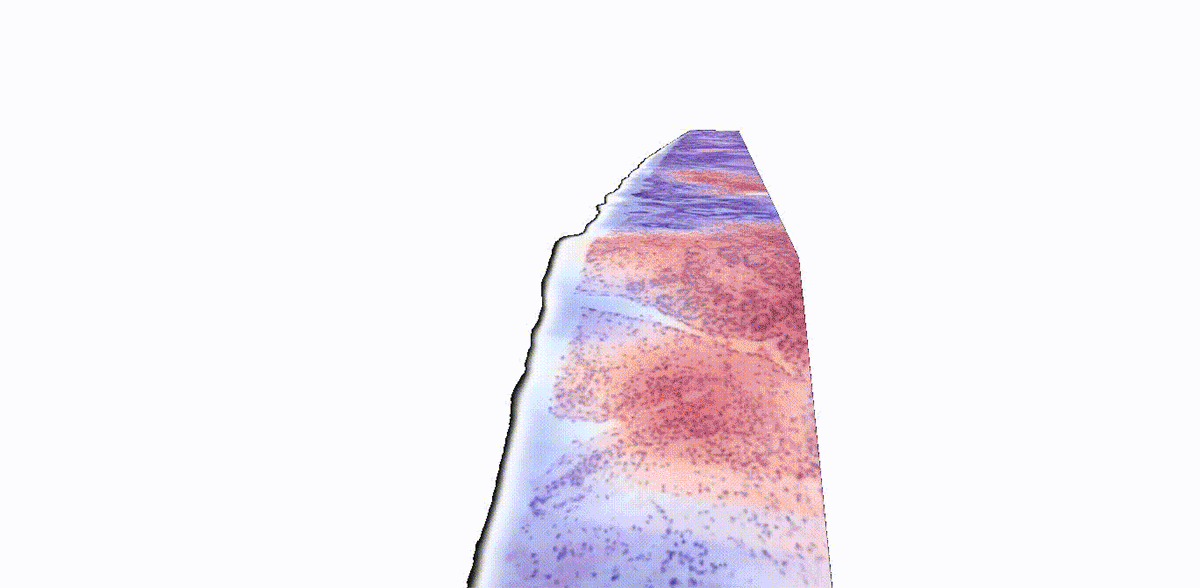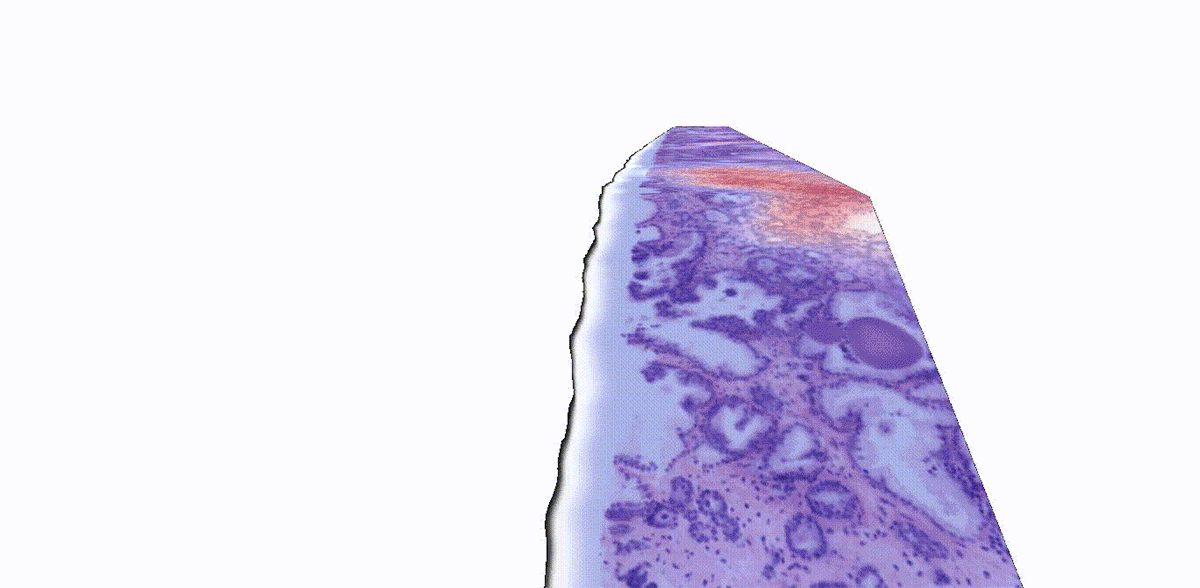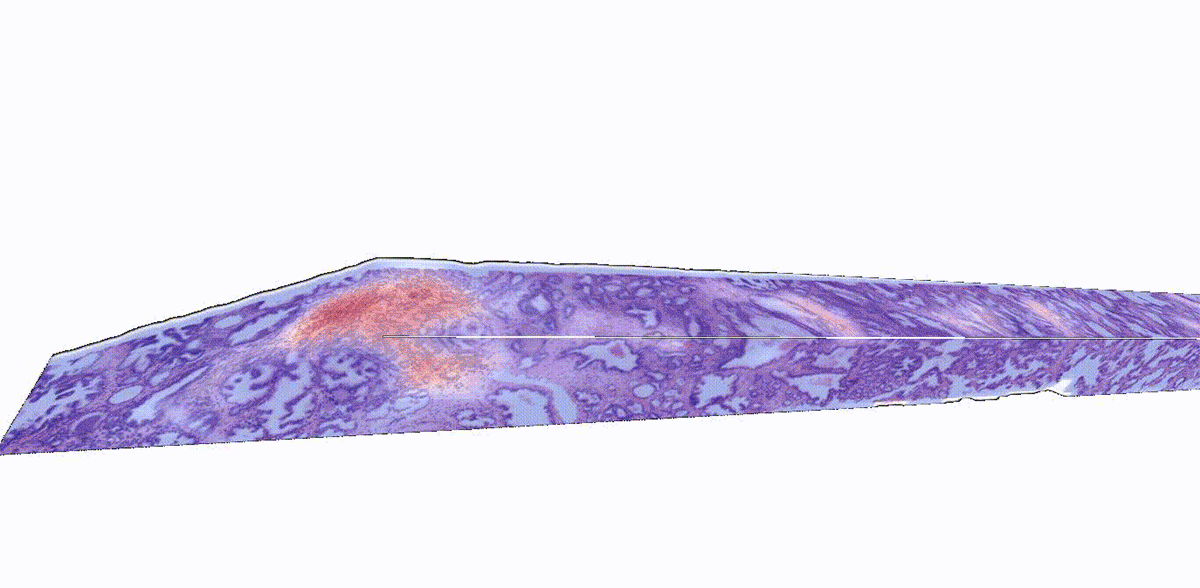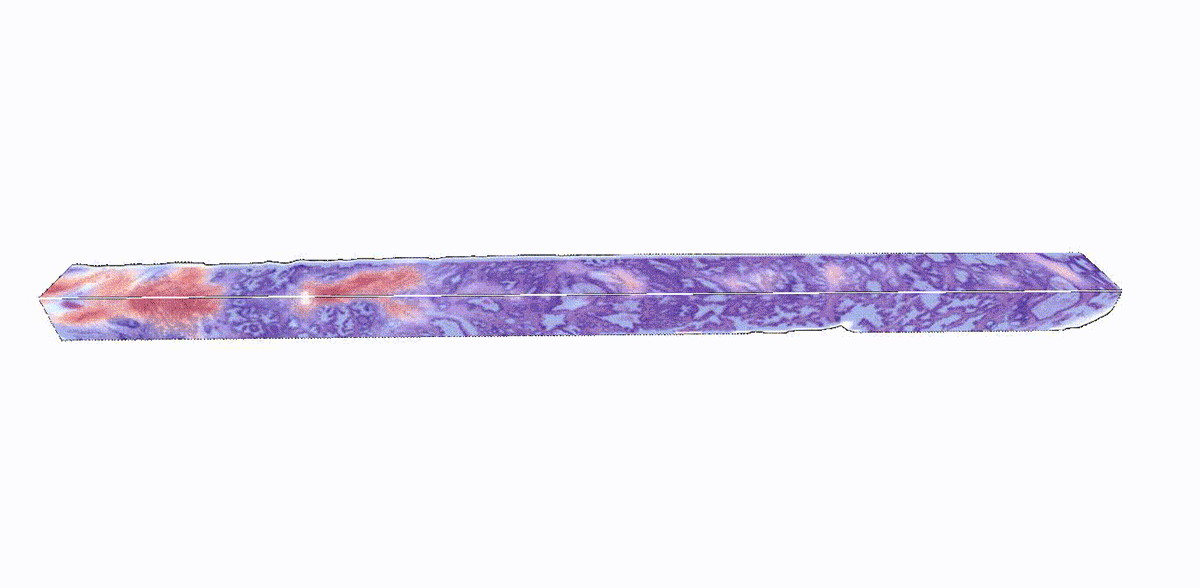
Adding another dimension to tissue analysis: The analysis of human tissue samples hasn’t fundamentally changed for more than a century. This technique is inherently limited by its two-dimensional nature: 2D images can only provide information about a slice of tissue, which doesn’t represent a holistic view of the sample. While 3D pathology techniques have recently emerged, a major challenge is analyzing these enormously complex images, which are nearly impossible to evaluate manually. NIBIB-funded researchers have developed an AI platform called TriPath that can analyze 3D pathology images to predict disease outcomes. The researchers found that TriPath had improved performance in predicting prostate cancer outcomes when compared with traditional pathology approaches, such as analysis by expert pathologists using 2D images. While 3D images are not routinely analyzed in the clinic today, the researchers believe that their technology has the potential to augment 2D pathology approaches for multiple diagnostic tasks to ultimately improve patient outcomes.
OCT for ear infections: More than 80 percent of children will have at least one ear infection by their third birthday. These infections are characterized by the buildup of fluid behind the eardrum, but standard techniques don’t allow clinicians to see into the middle ear cavity. Diagnoses can therefore be somewhat subjective, and incorrect conclusions can lead to unnecessary antibiotic use or can prolong the time to necessary surgery. In response to this need, NIBIB-funded researchers have developed an OCT system that can generate cross-sectional images of the eardrum and the middle ear cavity, allowing clinicians to better determine whether ear-related pain requires immediate interventions. Their system, which was cleared by the FDA in 2022, was shown to reduce antibiotic use by 50% in an interim analysis of an ongoing randomized controlled trial that is comparing this OCT technology with standard of care.
Building the microscopes of tomorrow: Microscopes are among the most indispensable tools that biomedical researchers use to image and analyze the complex machinery of life. As new imaging technology emerges, it broadens the information that can be captured from living systems and drives a deeper understanding of biology. The Advanced Imaging and Microscopy (AIM) resource at NIH brings together engineers, biologists, and computer scientists to build new microscopes and computational algorithms that are designed to have a transformative impact on the study of health and disease. In a recent study, NIH researchers, with the assistance of AIM, generated 3D tissue maps at sub-cellular resolution to better study how different tissue types interact with implanted biomaterials. This research could shape the development of future implants to ensure that they are effective and safe.
Updated September 2025
