The microscope is a workhorse in laboratories for biomedical research and diagnosing disease from tissue samples such as tumor biopsies. A research team led by engineers at the University of Washington has developed a microscope that combines rapid high- and low-power light-sheet microscopy with an open top design that allows for rapid imaging of a wide range of sample types [1].
The research was led by Dr. Adam Glaser while he was working in the laboratory of principal investigator Jonathan Liu, professor of mechanical engineering, laboratory medicine & pathology, and bioengineering at the University of Washington with collaborators from across the country and internationally. Dr. Glaser is now a senior scientist at the Allen Institute for Neural Dynamics in Seattle, Washington.
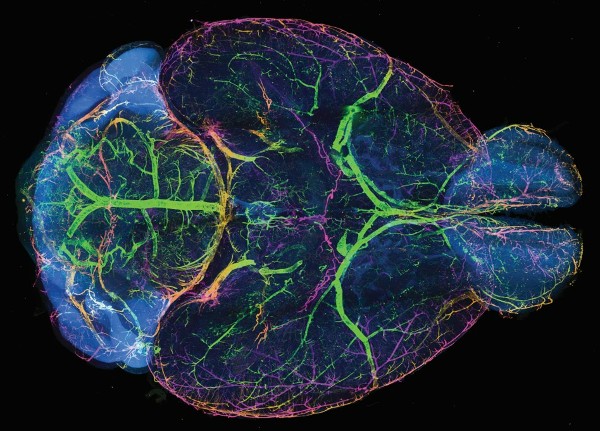
Dr. Liu’s laboratory has been focused on improving the utility of microscopy technologies for clinical applications and in research laboratories. Their new microscope is called a hybrid open-top light-sheet (OTLS) microscope, which brings characteristics of several different microscopes into one with the aim of reducing the scanning time and the computer resources necessary to efficiently and accurately analyze tissue specimens.
The microscope is unique in that it is “open-top.” Most microscopes have the lenses hovering over the top looking down at the sample, which severely limits the size of samples that can be placed on the platform just below the lenses. In the open top configuration, the lenses are underneath, “looking up” through the glass platform, which leaves the platform completely wide open so that large pieces of tissue or samples arrayed in the wells of culture plates can be easily placed on the platform and imaged.
The other innovation is that rather than a single objective found in most microscopes, the hybrid microscope contains three. One that sends a sheet of light into the sample to illuminate fluorescent dyes within the sample, and two that capture the fluorescent images that are generated by the illumination light sheet. The two image-capturing objectives allow the researcher to seamlessly view samples in both high and low power. When numerous samples need to be viewed—with the researcher or technician deciding whether to investigate a specific region more closely—the ability to rapidly view low and high power significantly streamlines what can be an arduous process.
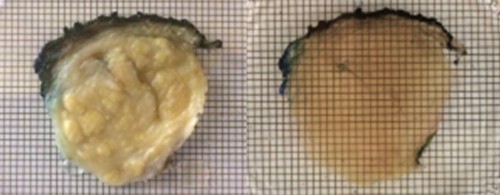
“The team has been cognizant of designing a microscopy system that significantly increases imaging efficiency at multiscale while also being accessible to multiple research and pathology laboratories,” said Afrouz Anderson, director of the program in Optical Imaging and Spectroscopy at the National Institute of Biomedical Imaging and Bioengineering, which co-funded the project. “The system is largely built from off-the-shelf parts, which will allow other labs to build and integrate it into their workflow.”
The research team demonstrated the utility of the microscope by imaging structures in whole mouse brains that were infused with what are known as clearing agents. Without the clearing step, images are distorted because light is scattered differently through different parts of the tissue. The clearing agent adds uniformity to the tissue, which reduces light scatter and produces significantly clearer images.
In one example, the microscope was used to obtain 3D fluorescent images of the nerve cells or axons in whole mouse brains. The hybrid system was able to quickly scan the entire brain at low resolution to identify target neurons which were then imaged with the high-resolution lens. A cubic millimeter region of the brain was imaged at high power to reveal the spines of individual neurons in just a little over an hour—a process that would have taken weeks of imaging with traditional microscopes as well as terabytes of imaging data versus the 180-gigabyte data set generated by the hybrid system.
In another experiment, the team imaged mouse brains containing metastatic tumor colonies formed by two types of human breast cancer cell lines. The open top design of the hybrid microscope allowed the research team to scan six-well tissue culture plates with a whole mouse brain in each well. The six-brain set could be rapidly scanned with the low-power objective to identify regions containing metastatic colonies. The high-power objective was then used to image and analyze the structural details of individual tumor colonies.

The detailed high-powered images allowed the researchers to observe that the two breast cancer cell lines formed different types of cancer cell colonies—one growing in a dense mass with clearly formed blood vessels feeding the cancer cells while the second cancer cell line created more diffuse colonies without clear attachments to the blood vessels in the brain.
The brain metastasis study was performed to replicate a study that had been previously published by another research team, which required manual transfer of specimens between two separate microscope systems and several weeks of tedious imaging to complete. By contrast the hybrid system allowed the entire imaging experiment to be completed in about two days—three to four hours of imaging each brain at low power and less than an hour to image individual metastatic colonies.
“The dedication of Adam and the rest of our colleagues has really paid off with an imaging system that promises to significantly improve both research and disease diagnosis,” explains Liu. “Used in a recent experiment aimed at the diagnosis of prostate cancer, open-top light-sheet microscopy was able to obtain detailed 3D images of whole prostate biopsies from patients. The detailed 3D images of the samples revealed structural differences in slow growing versus aggressive prostate cancer samples that we believe may guide improved treatment decisions for many men with prostate cancer [2].”
This work was funded by multiple sources (see published paper for a full list), including the National Institute of Biomedical Imaging and Bioengineering grant R01EB031002, the National Cancer Institute grant K99CA240681, and the Department of Defense Prostate Cancer Research Program.
-Written by Thomas Johnson, Ph.D.
1. A hybrid open-top light-sheet microscope for versatile multi-scale imaging of cleared tissues. Glaser AK, Bishop KW, Barner LA, Susaki EA, Kubota SI, Gao G, Serafin RB, Balaram P, Turschak E, Nicovich PR, Lai H, Lucas LAG, Yi Y, Nichols EK, Huang H, Reder NP, Wilson JJ, Sivakumar R, Shamskhou E, Stoltzfus CR, Wei X, Hempton AK, Pende M, Murawala P, Dodt HU, Imaizumi T, Shendure J, Beliveau BJ, Gerner MY, Xin L, Zhao H, True LD, Reid RC, Chandrashekar J, Ueda HR, Svoboda K, Liu JTC. Nat Methods. 2022 May;19(5):613-619. doi: 10.1038/s41592-022-01468-5. Epub 2022 May 11. PMID: 35545715
2. Prostate Cancer Risk Stratification via Nondestructive 3D Pathology with Deep Learning-Assisted Gland Analysis. Xie W, Reder NP, Koyuncu C, Leo P, Hawley S, Huang H, Mao C, Postupna N, Kang S, Serafin R, Gao G, Han Q, Bishop KW, Barner LA, Fu P, Wright JL, Keene CD, Vaughan JC, Janowczyk A, Glaser AK, Madabhushi A, True LD, Liu JTC. Cancer Res. 2022 Jan 15;82(2):334-345. doi: 10.1158/0008-5472.CAN-21-2843. Epub 2021 Dec 1. PMID: 34853071
