Photoacoustic cancer imaging in mice paves way for use in humans
1. Uses ultrasound device found in most health care settings and inexpensive laser light source.
2. No ionizing radiation allows for repeat imaging to monitor tumor response to treatment.
3. Nanoparticle contrast agent can be engineered to target different tumor types.
High-quality PA images can be obtained using contrast agents that target specific molecules such as receptors commonly overexpressed by cancer cells. The contrast agents are highly light-sensitive molecules that are infused systemically into a patient and bind to the tumor surface. When exposed to pulsed light, the contrast agent creates minute vibrations that can be detected by ultrasound to provide molecular images suitable for clinical applications such as tumor detection.
To date, the only clinically approved imaging system that is also capable of detecting molecular changes in tissues and organs is positron emission tomography (PET), which uses ionizing radiation and complex and expensive machinery, which limits its use for widespread and repeated cancer imaging. Ionizing radiation can cause damage to tissue, especially if a patient is exposed repeatedly.
Because it does not use ionizing radiation and the equipment needed is much less expensive than PET imaging devices, PA imaging is poised to fill an “imaging gap.” That gap exists in clinics and healthcare facilities that do not have expensive PET imagers, but often already have the ultrasound machines needed for PA imaging. Such clinics, with the addition of an accessibly priced laser that generates the necessary pulses of light, would be poised to offer point-of-care cancer imaging to patients at local sites. In particular, PA imaging is ideal for patients undergoing cancer treatment as the non-ionizing technique can be performed repeatedly to monitor a tumor’s response to therapy.
Unfortunately, this promising imaging system has been slow to move from the laboratory into the clinic. To address this problem, researchers at the University of Texas MD Anderson Cancer Center are working to develop a molecular contrast agent for PA imaging that is ready for the clinic and has potential to detect breast, ovarian, prostate, and other tumor types.
“This research was driven by two important goals,” explained Richard Bouchard, in the MD Anderson Department of Imaging Physics and co-senior author of the study. “First, design and synthesize a nanoparticle contrast agent with all of the characteristics necessary for it to generate a high-contrast image of cell receptors overexpressed by cancer cells at clinically relevant depths, which would be centimeters beneath the skin. Second, build that nanoparticle entirely out of components that are already approved by the FDA to avoid regulatory hurdles that have significantly hampered getting this technology into the clinic.”
The necessary characteristics of the nanoparticle envisioned by Bouchard and colleagues included the ability to reach and bind to molecules of interest, such as specific proteins found on certain tumors. The particle also had to optically absorb enough for the PA signal to be strong enough to bounce back through the body and skin and be clearly detected by an ultrasound imaging detector. The optical absorbance had to occur when a narrow wavelength of near infrared light was used to avoid non-specific stimulation of surrounding tissues that create noisy, blurry images. Finally, the individual parts of the nanoparticle had to be previously approved for human use by the FDA.
“This is a comprehensive effort that brings together expertise in chemical synthesis, nanotechnology, photonics, and ultrasound technologies,” said Tatjana Atanasijevic, Ph.D., who manages the program in Molecular Probes and Imaging Agents at the National Institute for Biomedical Imaging and Bioengineering (NIBIB), which co-funded the project along with several additional institutes at the National Institutes of Health. “The work goes a long way towards moving this valuable technology closer to use in the clinic.”
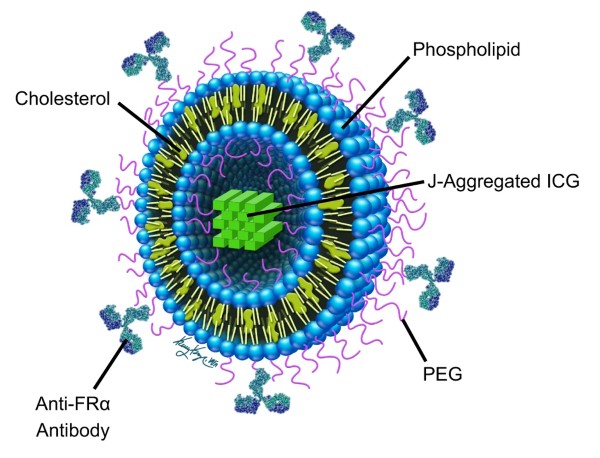
A dye called indocyanine green (ICG), which is already used in humans for imaging, was selected as the contrast agent housed in the center of the particle. However, ICG creates a relatively weak signal, so the team synthesized an aggregate of multiple ICG molecules. Such aggregation results in increased PA signal generation for deeper tissue imaging.
In addition, the ICG aggregate generates a strong PA signal at a narrow NIR wavelength of 890 nanometers, which avoids the problem of confounding PA signals being generated by surrounding tissues, most notably hemoglobin in the blood. Because blood vessels permeate all organs and are the conduit for systemic delivery of such nanoparticles, it was critical that their agent could be reliably differentiated from hemoglobin to obtain high-contrast images.
To test the nanoparticle, the team used a mouse model of ovarian cancer. Getting the nanoparticle, introduced systemically, to home to and bind to the ovarian tumors was achieved by adding an antibody to the surface of the particle. The antibody specifically binds to a molecule overexpressed on the surface of ovarian tumors called the folate receptor.
Finally, all the parts were packaged in tiny nanospheres built with cholesterol and other lipids already found in the body and used routinely for drug delivery. The completed “stealth” molecule was named PAtrace.
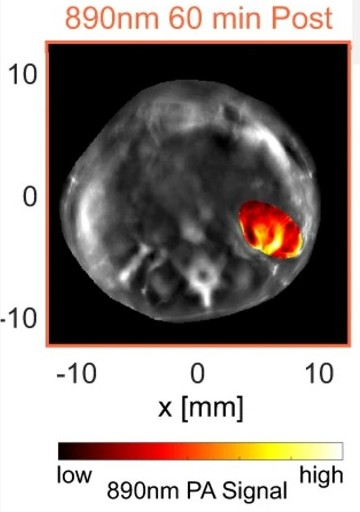
PAtrace was tested in a mouse model of ovarian cancer, which contains human ovarian cancer cells that have been seeded into the mouse ovaries. The nanoparticle was infused into the circulation of the mouse where it flowed to the ovaries and was small enough to escape from the vasculature and bind to nearby ovarian cancer cells. PA imaging was conducted and a clear image of the BB-sized tumors on the mouse ovaries was generated by PAtrace. As designed, the hemoglobin in the surrounding blood vessels did not “light up,” allowing for a clear image of the tumors.
“Our testing of PAtrace revealed that the components we chose functioned extremely well in terms of the various parameters necessary to make this type of imaging a viable option,” explained Konstantin Sokolov, from the MD Anderson’s Department of Imaging Physics and co-senior author of the study. “We are now further testing PAtrace synthesized with new targeting molecules on the surface of the nanoparticle that will home to and bind to tumors in various organs such as breast. The ability to “swap out” the targeting molecules on the surface add a versatility to the system as we work toward eventual use in humans.”
This work was supported by grant R01EB028762 from the National Institute of Biomedical Imaging and Bioengineering, the National Cancer Institute, the Office of the Director of the National Institutes of Health, the American Cancer Society, the Frank McGraw Memorial Chair in Cancer Research, the Sao Paulo Research Foundation, and the Cancer Prevention & Research Institute of Texas. The manuscript appears in the September issue of Nature Communications1.
—Thomas Johnson, Ph.D., special to NIBIB
