NIBIB’s HEAL grantees develop a new type of injectable electrode for neuromodulation therapy
Researchers at the University of Wisconsin (UW) are adapting a minimally invasive, safer approach to electrically treat pain directly at the source as part of the Helping to End Addiction Long-term InitiativeSM, or the NIH HEAL InitiativeSM. It is an advance from one of the National Institute of Biomedical Imaging and Bioengineering’s (NIBIB’s) first grants under the NIH HEAL InitiativeSM, launched in 2018 to find solutions to the opioid crisis.
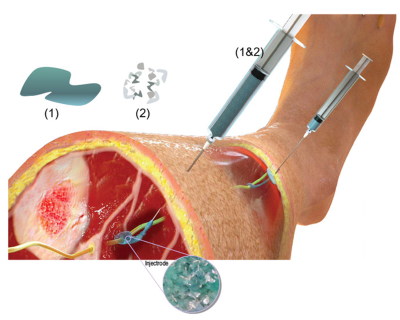
Currently, the most effective neuromodulation treatments require intricate surgical procedures to implant a complex device that is invasive and expensive, making it a treatment of last resort. In contrast, non-invasive neuromodulation devices apply stimulation through electrodes that are placed on the surface of the skin and use a specially crafted electrical waveform to block painful signals in nerves. These electrical waveforms travel through tissue, muscle, and superficial nerves to achieve a therapeutic effect. Unfortunately, surface electrodes can result in uncomfortable sensations due to undesired stimulation of the superficial nerves. This approach has limited effectiveness for target nerves that are deeply buried under the skin.
“To address these limitations, the Wisconsin team has created a new technique that has the effectiveness of an implantable device and is only marginally more invasive than surface electrodes,” said Michael Wolfson, Ph.D., director of the NIBIB Program in Bionic and Robotic Systems. By using an injectable liquid polymer that cures – like household epoxy -- next to or around the target nerve, the researchers say the device provides a localized stimulus that avoids the side effects of other types of neuromodulation therapy. The new liquid polymer electrode named “Injectrode” is made of softer materials than traditional implantable devices, and is mechanically similar to human tissue, allowing it to integrate more easily, all while providing electrical conductivity similar to a metallic wire.
The UW-Madison team, led by Kip Ludwig, a professor of biomedical engineering and neurological surgery, presented their work in Advanced Healthcare Materials, showing that the Injectrode could form flexible structures around a variety of pig nerves and provide an electrical stimulus to a pig’s vagus nerve. The vagus nerve serves as the critical communication link between the brainstem and the rest of the body, particularly organs and tissues. Vagus nerve stimulation therapy has also been used for treating depression and epilepsy. The group reported that their technology had passed several preclinical safety standards recognized by the Food and Drug Administration for this type of device.
“This is an innovative approach to interfacing with the nervous system, and I’m excited to see their HEAL grant push hard towards first-in-human demonstrations,” said Wolfson. “Even in the midst of a pandemic, the opioid crisis continues unabated, and this new technology offers hope to individuals suffering from debilitating back pain.”
Collaborators at the University of Michigan are developing computational simulations to optimize the delivery of current to a cluster of neurons in the spinal nerve called the dorsal root ganglia.This cluster of nerves are a primary target for neuromodulation therapies Case Western Reserve University collaborators are characterizing and optimizing the mechanical properties of the Injectrode. Lastly, tying it all together on the Ludwig team is the preclinical work conducted by the University of Pittsburgh researchers who are performing efficacy testing to aid in the translation process of bringing this pain treatment to market. Neuronoff, Inc., and Ludwig’s team will continue to develop the Injectrode with further safety and effectiveness experiments, as well as techniques to reliably administer it and power it with a wireless stimulator.
The work was supported by the NIBIB (1U18EB029251‐01). This work was also supported by The Defense Advanced Research Projects Agency (DARPA) Biological Technologies Office (BTO) Targeted Neuroplasticity Training Program under the auspices of Doug Weber and Tristan McClure‐Begley through Space and Naval Warfare Systems Command (SPAWAR) Systems Center with (SSC) Pacific grant no. N66001‐17‐2‐4010 and The Grainger Foundation.
James K. Trevathan, et al., “An Injectable Neural Stimulation Electrode Made from an In‐Body Curing Polymer/Metal Composite,” Advanced Healthcare Materials, 2019.
