Preclinical study evaluated treatments for diabetes, Crohn’s disease, and other conditions that use injectable meds using an orally administered capsule
Biologic drugs—so named because they are typically isolated from a living source, rather than chemically synthesized—are used to treat a wide variety of conditions, including diabetes, inflammatory diseases, and certain types of cancer. However, due to their complex and easily degradable components, the administration of these drugs often requires a self-injection, which can represent burdens for patients, such as necessary training for proper self-administration and the potential for needle stick injuries and pain. Now, NIBIB-funded researchers are developing a robotic pill that, after swallowing, can deliver biologic drugs into the stomach, which could potentially revolutionize the way that certain conditions are treated.
“Due to the inherent drawbacks of injectable medications, many healthcare professionals prescribe less effective oral medications in their place, resulting in suboptimal treatment for many patients,” said David Rampulla, Ph.D., director of the division of Discovery Science & Technology at NIBIB. “An oral pill for the delivery of biologic drugs would not only positively impact those patients who already use injectable medications, but could also benefit patients that are currently delaying their use. This preclinical research is an important step toward the development of such an approach.”

The study authors built on previous research where they developed robotic pills that were capable of delivering solid biologic drugs into the systemic circulation via injection into the stomach and small intestine. These devices, evaluated in animal models, accommodated lower drug doses (around half a milligram per capsule), which would necessitate frequent oral dosing for some commonly used biologic drugs. They also had limited immediate drug absorption, preventing their use for fast-acting purposes (such as mealtime insulin). “We applied lessons learned from our previous work and developed a new device that can inject a hollow needle into a shallow layer of the stomach lining, which allows for the delivery of liquid drugs,” explained senior study author Giovanni Traverso, M.D., Ph.D, assistant professor in the Department of Mechanical Engineering at Massachusetts Institute of Technology. “The delivery of a liquid formulation enables drug uptake and effects within five minutes following capsule ingestion,” noted Traverso, who is also an associate physician at Brigham and Women’s Hospital and an assistant professor at Harvard Medical School.
He explained how their improved device works in a domino-type fashion: Once the pill has been swallowed and makes its way into the stomach, it uses its weighted bottom to orient itself properly, so that its injection mechanism is flush against the stomach wall. After a few minutes, a carbohydrate pellet at the top of the pill dissolves, activating a spring and enabling a needle to inject the biologic drug into the stomach tissue. Then, a second, newly exposed pellet dissolves, freeing the spring and retracting the needle back inside the pill, allowing for safe passage of the device through the gastrointestinal tract.
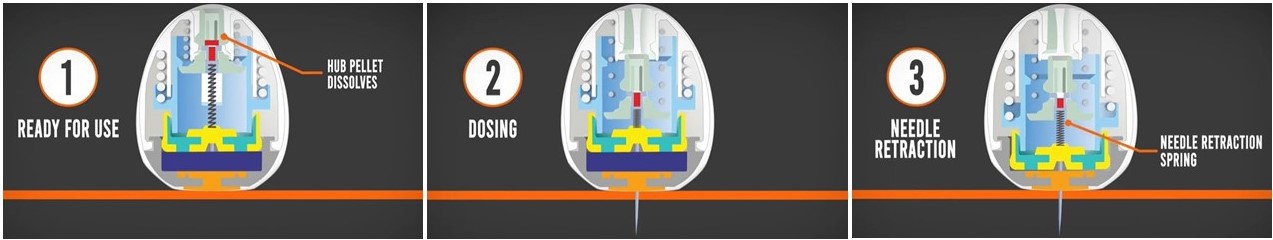
“For the shape of our robotic pill, we were inspired by weeble-wobble toys and leopard tortoises, which both have a wide, heavy bottom that allows them to self-right themselves,” said study author Ulrik Rahbek, Ph.D., vice president of Partnerships and Portfolio in Research & Early Development at Novo Nordisk, a Danish multinational pharmaceutical company. “This ensures that the biologic drug is successfully delivered into the stomach tissue rather than into the stomach cavity, which is filled with enzymes that can easily degrade and inactivate these sensitive drugs.”
In their study, reported in Nature Biotechnology, the authors loaded their robotic pills with one of four biologic drugs—insulin; a glucagon-like peptide 1 (GLP-1) analog (also used for the management of diabetes); adalimumab (brand name Humira®, an immunosuppressive drug used for a variety of conditions, including arthritis and Crohn’s disease); or epinephrine (for the emergency treatment of allergic reactions or asthma attacks). Using an endoscope, the researchers placed the drug-loaded robotic pills into the stomachs of swine and allowed them to self-activate. Then, after 15 minutes or two hours (depending on the drug), the pills were removed. The researchers evaluated drug exposure in the blood of the animals along with other relevant metrics (like hypoglycemic onset for insulin treatment or rise in heart rate for epinephrine treatment).
In the 31 large animals in this preclinical study, 28 demonstrated systemic uptake of the dosed biologic drug, resulting in a positive dosing rate above 90%.
To evaluate how well the robotic pills worked compared with traditional administrations of biologic drugs, the researchers gave separate pigs the same four medications, given as either subcutaneous (fatty tissue) or intramuscular injections. They found that treatment administration using the robotic pills resulted in similar drug exposures as treatment administration using traditional injection methods for all four of the evaluated drugs.
“We found that our device results in drug uptake that is comparable to current injection methods and can facilitate drug exposure within minutes,” noted Traverso. “Combined with the fact that our capsule can deliver large doses—up to four milligrams—of complex biological drugs, our robotic pill could have a transformative impact on patients who rely on injectable medications.”
In order to assess if the robotic pills could be ingested and passed through the gastrointestinal tract without difficulty, the researchers evaluated similar robotic pills (without any drugs) in eight dogs. They found that all the animals could ingest the capsule with ease, and radiographic monitoring revealed that the devices traveled to the stomach, activated, and then traveled through the gastrointestinal tract as expected.
The study authors stressed the preclinical nature of the current study, and that human testing has yet to be performed.
“While there is still much work to be done, this capsule has the potential to offer an alternative to injections across a range of therapies,” concluded Rahbek.
Traverso and colleagues are also investigating their robotic pills for the delivery of therapeutic nucleic acids, such as mRNA vaccines, in animal models. Early work was just published in Matter.
Studies referenced herein were supported in part by a grant from NIBIB (EB-000244).
Study reference: Abramson, A., Frederiksen, M.R., Vegge, A. et al. Oral delivery of systemic monoclonal antibodies, peptides and small molecules using gastric auto-injectors. Nat Biotechnol 40, 103–109 (2022). https://doi.org/10.1038/s41587-021-01024-0
