Fluid exchange during sleep may clear brain of toxic substances
Neurologists have observed reduced neural activity during non-rapid eye movement sleep (NREM). Now MRI imaging during NREM reveals an exchange between brain blood and cerebrospinal fluid that may function to remove neurotoxic waste products.
Researchers at Boston University simultaneously monitored neural activity and movement of blood and cerebrospinal fluid (CSF) in the brain during NREM. The surprising finding was that during the reduction in neuronal activity that accompanies NREM, blood flows out of the brain allowing CSF to enter—in the opposite direction that it normally circulates through the brain. The work was led by Laura D. Lewis, Ph.D., Assistant Professor in the Department of Biomedical Engineering at Boston University, in collaboration with co-authors at the Martinos Center at Massachusetts General Hospital. The findings were reported in the November issue of Science1.
It is known that a good night’s sleep promotes physical and mental health. Good sleep is characterized by a rhythmic cycle of entering shallow NREM, moving into deep NREM, moving back to shallow NREM, and finally into rapid eye movement (REM) sleep before the cycle repeats.
Deep NREM is essentially a resting state of brain neurons characterized by slow rhythmic waves of electrical activity quite different from the more complex neural activity observed during various physical and mental tasks.
“Based on the known correlation between good sleep and good health, the researchers set out to find physiological mechanisms that might explain that observation,” said Shumin Wang, Ph.D., director of the NIBIB program in Magnetic Resonance Imaging and Bioelectromagnetics. “They combined electroencephalography (EEG), which measures the electrical activity of the brain, with functional MRI, which monitors changes in blood flow, to identify a mechanism that could explain how sleep promotes rest and renewal of the brain and how neurodegenerative disease may be tied to disruption of that process.”
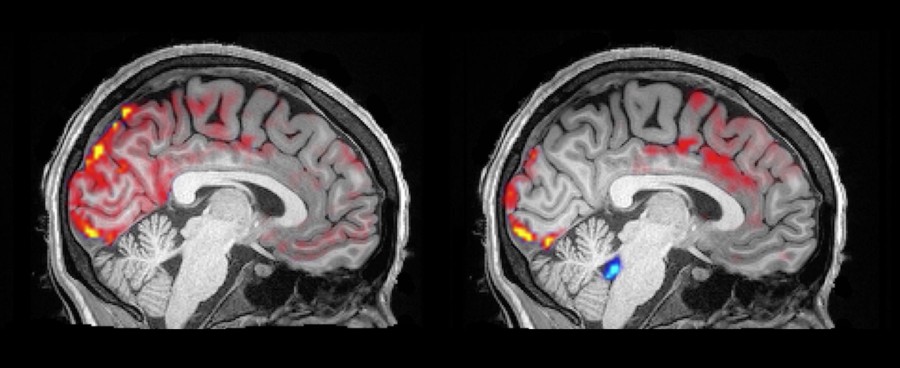
Lewis explained the rather difficult experimental set-up. “We had individuals wearing (EEG) caps, while lying motionless in a noisy MRI machine, trying to sleep.” She noted that the 13 volunteers ages 23-33 loved the idea of getting paid to sleep but found falling asleep tougher than they had expected in the uncomfortable, noisy environment. The experimental set-up allowed the researchers to monitor the brain’s electrical activity and the movement of CSF and the blood in the brain, simultaneously.
What they found was that when the volunteers entered NREM and their brain neurons switched to a slow rhythmic pattern, the volume of blood in the brain decreased, leaving open spaces in the brain that then filled with CSF. The movement of the CSF into those spaces was far larger and faster than the CSF flow that normally occurs during the day.
The researchers believe this movement of CSF could contribute to a rinsing of the brain, which is likely to remove toxic waste products that build up during the day.
The results are particularly exciting because it is known that NREM declines as people age, and even more dramatically in those with neurogenerative diseases such as Alzheimer’s. The research team plans to look in older adults, who have reduced sleep quality, to see if they have reduced periods of NREM as well as reduced pulsing of CSF into and out of the brain during NREM.
Ultimately, the protocol might someday become a diagnostic test that identifies individuals with higher risk of neurodegenerative diseases including Alzheimer’s.
The work was supported by Grants EB019437, EB015896, and EB024343 from the National Institute of Biomedical Imaging and Bioengineering, the National Institute of Mental Health, the National Institute of Neurological Disorders and Stroke, the NIH Office of the Director, and the Martinos Center for Biomedical Imaging.
